Advanced Materials for More Efficient PEM Electrolysis
The discussion around hydrogen from renewable energy sources often focuses on infrastructure, costs, and scalability. The structure of the electrolysis cell underlying the technology, often receives much less attention. Yet every component, from membrane to electrode to catalyst, affects the overall system. In the Bosch Hydrogen Expert Talk, Anna Bernroitner, electrochemistry expert at the Bosch site in Linz, explains how materials science considerations can play a decisive role in optimizing the electrolysis stack.
-253°C | The Bosch Hydrogen Expert Talk #9: wH₂y material matters

Loading the video requires your consent. If you agree by clicking on the Play icon, the video will load and data will be transmitted to Google as well as information will be accessed and stored by Google on your device. Google may be able to link these data or information with existing data.
In this episode, Anna Bernroitner, electrochemistry expert at Bosch, explains how she and her team significantly contribute to optimizing the electrolysis stack through materials science considerations.
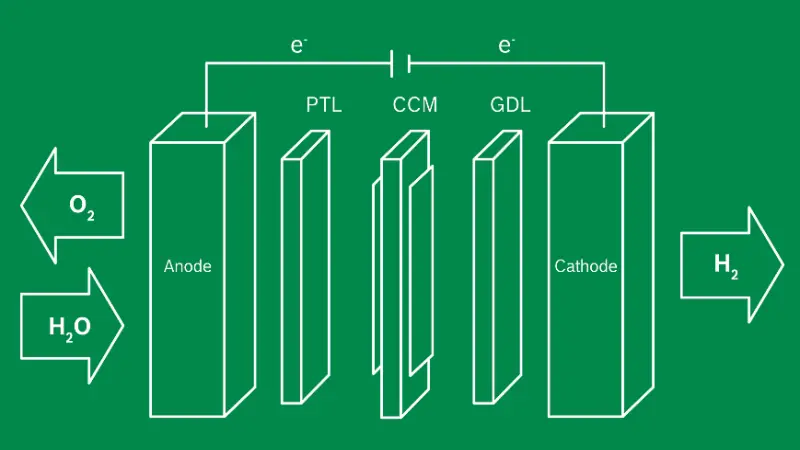
What happens during PEM electrolysis?

PEM electrolysis (Proton Exchange Membrane Electrolysis) is a process that splits water into hydrogen and oxygen using electricity.
At the anode, water molecules is decomposed, while hydrogen gas is generated at the cathode.
Electrolysis – a challenge in materials science

To understand and deliberately improve electrochemical systems, we study and investigate the properties of the materials as precisely as possible. For this purpose, we conduct electrochemical tests in full scale and sub scale stacks, complemented by simulations of model systems.
With the CCM, we can perform tensile and compression tests to understand how mechanical stresses affect performance and durability. The knowledge gained can be used in simulations, which in turn promote new insights. This creates a continuous cycle of research, understanding, and innovation.
How do we optimize the durability of the PEM electrolysis stack?
One example is the compression of the CCM, as it plays a crucial role in the performance and durability of the system. Stronger compression improves the electrical contact between PTL, GDL, and the CCM, which leads to higher efficiency. At the same time, the mechanical stress increases, which can accelerate aging or hinder the flow of materials to and from the reaction sites. Our goal is to find the optimal balance that ensures a long lifespan without compromising performance.
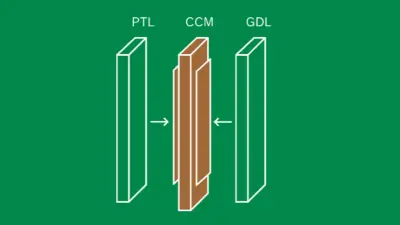
How can we better understand the aging mechanisms of the membrane?
Over time, the CCM is subject to various aging processes, both mechanical and chemical. Mechanically, the membrane can become thinner due to constant compressive stress. Another possibility is chemical membrane degradation, where the CCM loses some of the polymer molecules that make up the membrane. To better understand these processes, we deliberately contaminate the electrolysis cell with various ions that can act catalytically. Some of these ions accelerate chemical degradation processes, while others block the important sites within the polymer that are necessary for proton conduction. The catalysts on the outside of the CCM can become ineffective over time. For instance through catalyst poisoning in which the active sites are blocked. Understanding these interactions is crucial for developing membranes that remain stable and efficient over the long term.